C Single Bond O Ir
Most alkyne CC bonds have stretching frequencies between 2100 and 2200 cm-1. This is due to the CO 2 in the beam poor background correction.
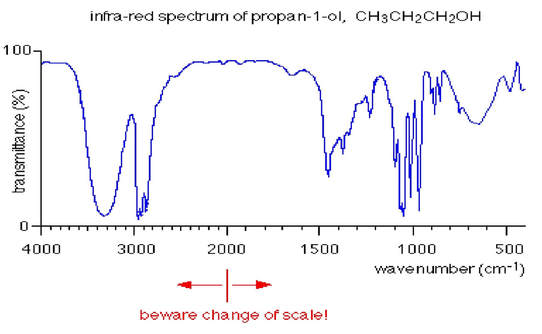
Science Skool Infrared Spectroscopy
EXC N O This is the most important range in the entire IR spectrum for organic chemists.
C single bond o ir. O-H 2500-3300 cm-1 very broad strong CO 1710-1760 cm 1 dimers lower monomers higher Amides. The change in dipole moment is significant making this an intense band and there are few other groups that give rise to absorbances in the 1600-1850 cm-1 range. As a result the carbon in the CO bond of aldehydes is also bonded to another carbon and a hydrogen whereas the same carbon in a ketone is bonded to two other carbons.
In ethane the orbitals are sp3-hybridized orbitals but single bonds formed between. C-S and S-H stretch stronger in Raman S-S stretch not visible in IR or Raman Strong P-H stretch 2440-2275 cm-1 PO stretch 1320-1140 cm-1 Only see P-O stretch when R is small alkyl groups in P-O-R OP-OH broad IR bands involving OH stretch from 2725-1600 cm. This band is due to the highly polar CO bond.
The CO double bond has a large dipole moment and produces intense IR absorptions 1700cm-1. From 1500 - 2000 cm-1 E-X-double bonds. IR of Acids Amides Anhydrides and Acyl Halides.
Infrared absorption only occurs when the incoming IR photon. The C-O single bond stretch more polar occurs around 1100 cm-1. Bond in a carboxylic acid has a partial double-bond character that is due to resonance electron donation.
The CO stretches appear as two or more bands in the region 1300-1000 cm -1. The C-N-triple bond of nitriles example 10 2100-2160 cm-1. You have to be very wary about picking out a particular trough as.
Despite being in the fingerprint region the C-O single bond stretch is exceptionally useful. The carbonoxygen single bond in a carboxylic acid has partial double bond character. Electron density between C and O.
IR peaks are not 100 reliable. Carboxylic Acids contain a carboxylic acid group a carbonyl CO group bonded to. Acyl All IR values are approximate and have a range of possibilities depending on the molecular environment in which the functional group resides.
As you increase the dipole moment the bond stretch frequency increases. A bond composed of two electrons one from each of the two atoms. Because of its position shape and size it is hard to.
The carbonyl stretch CO of aliphatic esters appears from 1750-1735 cm -1. Aldehydes and Ketones contain a CO carbonyl group note that in condensed structural formulas the aldehyde group may be written as CHO or as CHO I. Absorption of triple bonds eg.
Absorption of single bonds to hydrogen eg. Location of Peaks in an IR Spectrum Carbon Oxygen Single Bond Stretch CO The carbonoxygen bond in an alcohol is a pure single bond. The C-O stretch is of medium to strong intensity and appears in the 1000 1300 cm-1 region.
Other Key IR Concepts The Fingerprint Region. The higher the CO bond order the higher the frequency. Aldehydes and ketones show a strong prominent stake-shaped band around 1710 - 1720 cm-1 right in the middle of the spectrum.
Because CC bonds are stronger and stiffer than CC they absorb IR light at higher frequencies. For example the C-C single bond stretch occurs around 1000 cm-1. Carboxylic acid OH stretch.
N-H 3300-3500 cm-1 sharp medium varies with of Hs CO 1690 cm 1 in saturated amides. For electronegative atoms ie. Oxygen and nitrogen the peak will be broadened by intra or inter molecular hydrogen bonding.
Electronegativities and bond lengths. The hybridization of the carbon bonded to the oxygen has a large effect on the C-O stretching frequency. Terminal alkynes usually give sharp CC stretching signals of moderate intensity.
Furthermore the precise position can be readily correlated with resonance effects. The carbon-oxygen single bond also has an absorbtion in the fingerprint region varying between 1000 and 1300 cm-1 depending on the molecule it is in. Shortened single bonds are found with carboxylic acids 136 pm due to partial double bond character and elongated bonds are found in epoxides 147 pm.
The region of an IR spectrum between 1500 and 600 cm-1 is unique for every molecule so its called the fingerprint region. C-H N-H O-H occur around 4000-2500 cm-1. Obviously ketones and acids do not have these peaks.
Acyl All IR values are approximate and have a range of possibilities depending on the molecular environment in which the functional group resides. Functional Group Frequency cm-1 intensity. In most cases a peak with varying intensity around 2349 cm-1 together with 667 cm-1.
The CO bond is strongly polarized towards oxygen electronegativity of C vs O 255 vs 344Bond lengths for paraffinic CO bonds are in the range of 143 pm less than those of CN or CC bonds. IR absorption only occurs when IR radiation interacts with a molecule undergoing a change in dipole moment as it vibrates or rotates. The carbonyl group of an ester therefore has a C-O double-bond character than does the carbonyl group of a ketone so the former is stronger and harder to stretch.
Electron density between C and O. A carboncarbon bond is a covalent bond between two carbon atoms. The CC stretching absorption of an internal alkyne may be weak or absent.
One selection rule that influences the intensity of infrared absorptions is that a change in dipole moment should occur for a vibration to absorb infrared energy. That of α β-unsaturated esters appears from 1730-1715 cm -1. Of bonds bending of bonds or internal rotation around single bonds.
The most common form is the single bond. Resonance often modifies a peaks position because of electron delocalization CO lower acyl C-O higher etc. Absorption bands associated with CO bond stretching are usually very strong because a large change in the dipole takes place in that mode.
The CO group is one of the most easily recognized peaks in an IR spectrum. The carboncarbon single bond is a sigma bond and is formed between one hybridized orbital from each of the carbon atoms. The carbon-carbon bond has absorptions which occur over a wide range of wavenumbers in the fingerprint region - that makes it very difficult to pick out on an infra-red spectrum.
Aldehydes and Carboxylic Acids The CO bond is slightly stiffer than the CC bond and so absorbs at slightly higher frequency 1710cm-1. Resonance often modifies a peaks position because of electron delocalization CO lower acyl C-O higher etc. An aldehyde also shows a characteristic set of two C-H stretches at 2700 and 2800cm-1.
The carbonoxygen bond in an ether is a pure single bond. IR peaks are not 100 reliable.

Figure 4 Fourier Transform Infrared Spectroscopy Ftir Of Nanoparticles Scanning Electron Microscopy Fourier Transform Infrared Spectroscopy Zeta Potential
Infrared Absorbance Spectroscopy Ir Mendelset
How Can I Distinguish Functional Group Region And Fingerprint Region In A Infrared Spectrum Socratic
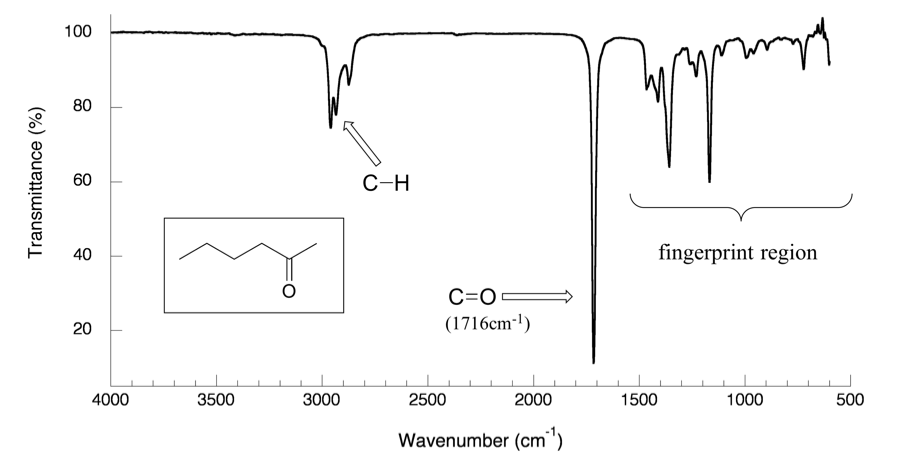
6 3 Ir Spectrum And Characteristic Absorption Bands Organic Chemistry
Infrared Absorbance Spectroscopy Ir Mendelset
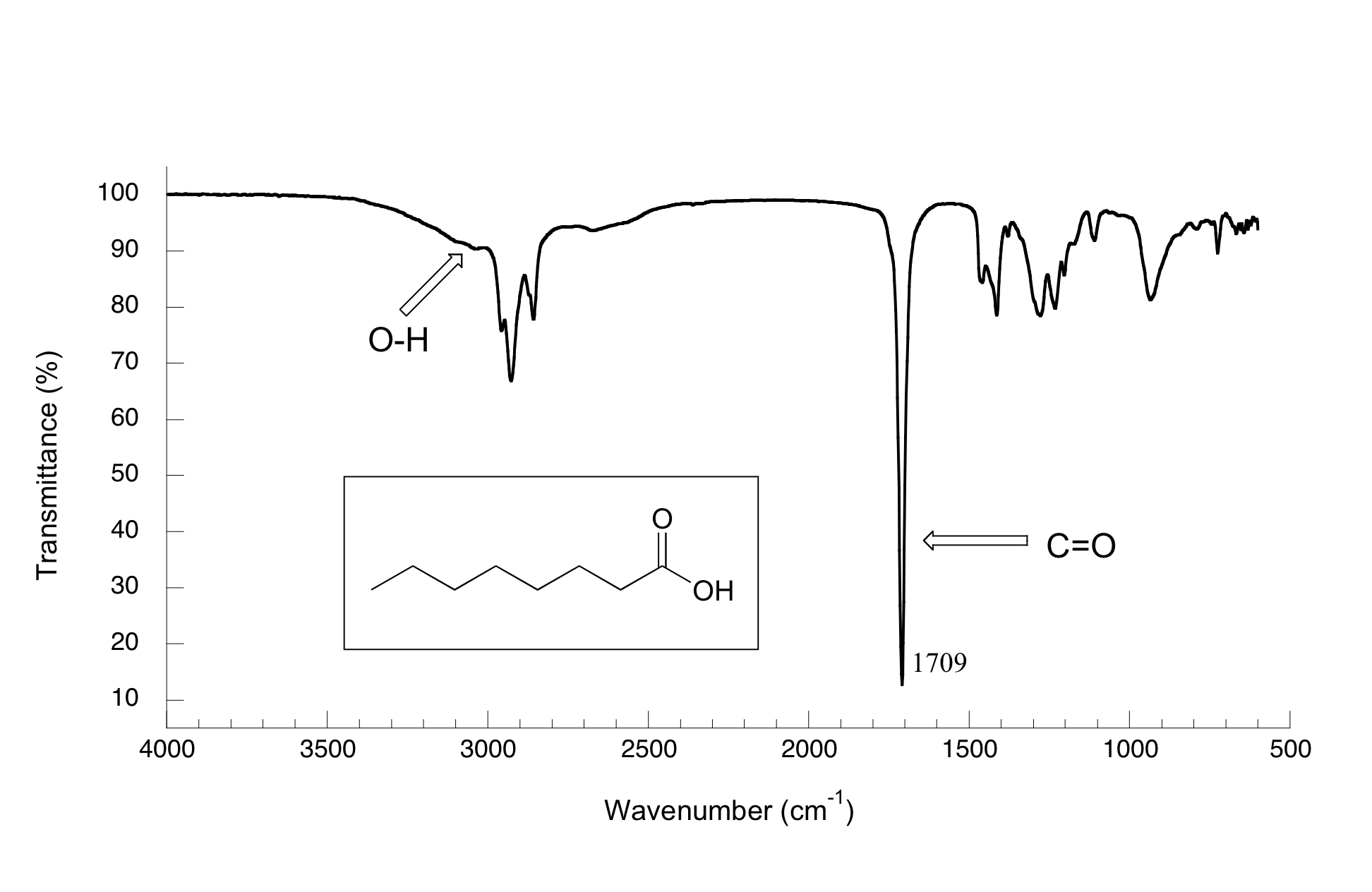
11 4 Interpretting Ir Spectra Chemistry Libretexts

Figure S3 Ft Ir Spectra For 3 3067 Cm 1 Aromatic C H Str 2225 Cm 1 Download Scientific Diagram
How Can I Distinguish Functional Group Region And Fingerprint Region In A Infrared Spectrum Socratic

Posting Komentar untuk "C Single Bond O Ir"