4 Single Covalent Bonds
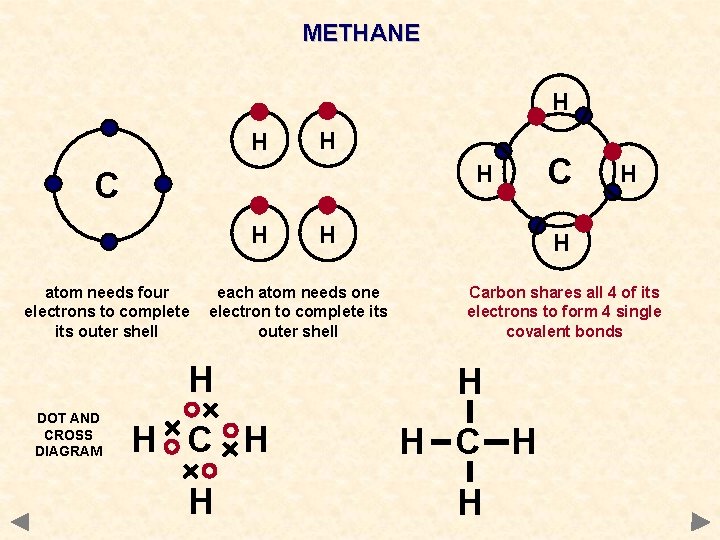
In structural formulas a single bond is represented by a line between bonded atoms. In the methane molecule the four hydrogen atom share one electron each with the carbon atom.
Covalent Bonding Covalent Bonding Definition Consists Of A
All 4 bonds in CH 4.
4 single covalent bonds. A carbon atom can form the following bonds. The type of covalent bonding in. A double covalent bond is formed when two pairs of electrons are shared between the two combining atoms.
1Single covalent bondIt is formed by sharing of one pair of electron between two atoms. In one form or another the idea of atoms connecting to form larger substances has been with us for a long time. 1Iron displaces copper from a solution of copper saltThe reaction is determined as redox.
Consider the molecule carbon dioxide CO. Pictorially a methane molecule may be represented as in Figure. Thus an atom of carbon forms four single covalent bonds with four H atoms.
ICSE X Chemistry Chemical Bonding - Ionic Compounds and Covalent Compounds Give reason. Secondly what type of bond is found in ch4. Simple molecular substances and giant covalent structures have different properties.
Covalent bonds form between non-metal atoms. Each electron pair represents one single bond-forming a single covalent bond. A compound has formula H2YY denotes a non metal.
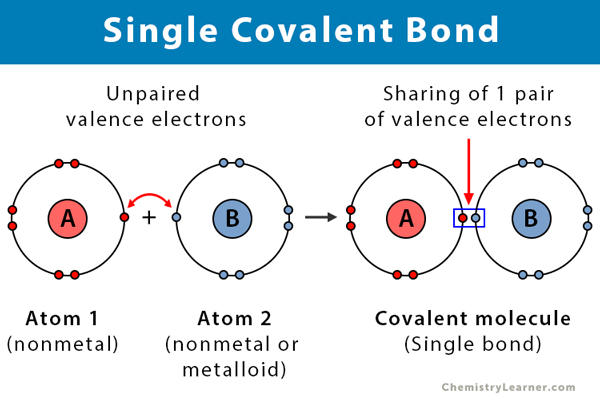
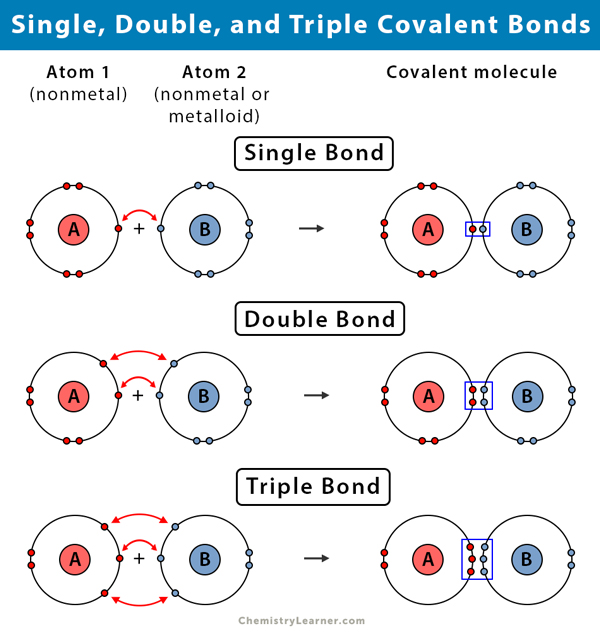
Single bonds are also seen in molecules made up of more than two atoms. A covalent bond forms when two non-metal atoms share a pair of electrons. A chemical bonding between the two atoms which shares a single pair of an electron is.
So if two identical nonmetals eg two hydrogen atoms bond together they will form a pure covalent bond. One short bond and one long bond Reality. A single covalent bond is one in which two electrons are shared by two atoms in which one electron comes from each atom.
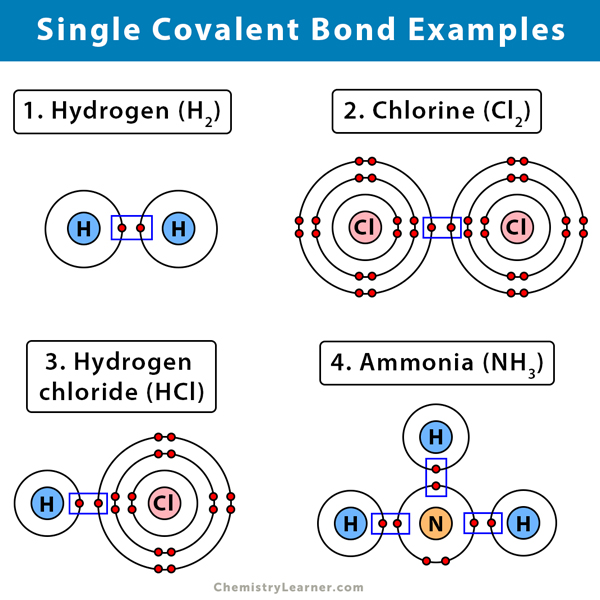
The Greek philosopher Democritus 460-370 BCE believed that atoms had hooks that allowed them to connect with one another. Examples of this use of single bonds include H 2 F 2 and HCl. Which of the following compound contains both polar and non-polar covalent bond.
2A non metaliic atom atno 9 forms a molecule of the samecontaining a single covalent bond. The structure of the methane CH4 molecule exhibits single covalent bonds. To the bonded atoms sharing the electron pair allows them to both feel like they have a complete compliment of electrons in their outer shell.
Both bonds in H 2 O. How many types of covalent bonds are there in class 10. Lesson Summary A chemical bond is.
Single bonding even appears in molecules as complex as hydrocarbons larger than methane. Oxygen has 4 electrons in covalent bonds. Each bond consists of a shared pair of electrons and is very strong.
A single covalent bond is when a single pair of electrons is being shared between two different atoms and there is one particular molecular orbital being occupied by that pair of electrons. Examples of this use of single bonds include. 2Double covalent bondIt is formed by sharing of two pair of electron between two atoms.
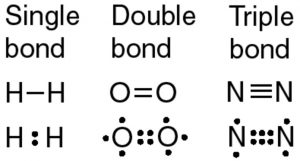
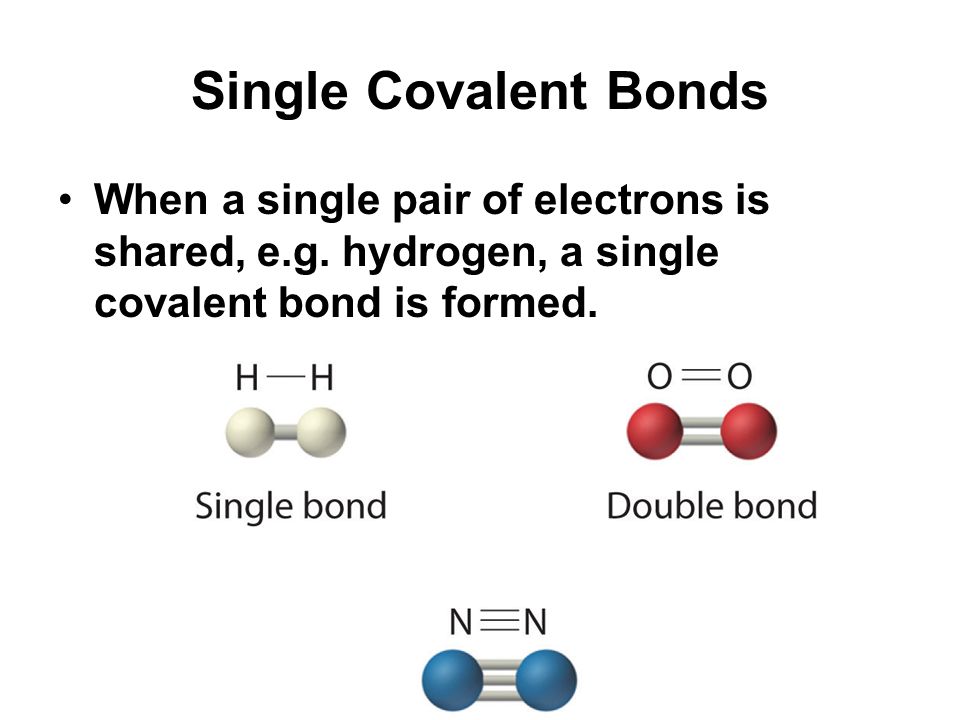
In Section 41 Covalent Bonds we stated that the covalent bond in the hydrogen molecule H 2 has a certain length about 74 10 11 m. Bonds are of equal length. Assign all 4 of these electrons to O.
Therefore the total number of electrons assigned. Carbon can form single double or triple covalent bonds with other carbon atoms. Diagram of single covalent bond being formed.
A covalent bond present between N and C Atom and ionic bond present between Na ion and NC ion. Covalent bonds form when two nonmetallic atoms have the same or similar electronegativity values. What Determines Whether a Covalent Bond Forms.
Table 42 Approximate Bond Lengths of Some Single Bonds lists the approximate bond lengths for some single covalent bonds. There are four single covalent bonds between carbon and hydrogen atoms resulting in a tetrahedral shape with a bond angle of 109. Single bonds are often seen in diatomic molecules.
One single bond Expect. B Single Bond. Covalent bonding involves the sharing of electrons.
Carbon has four valence electrons so it can achieve a full outer energy level by forming four covalent bonds. Since carbon atoms have four valence electrons they attain a stable electronic configuration by forming four covalent bonds with hydrogen atoms. Two single covalent bonds are present in the water molecule.
Assign 2 of these 4 electrons to O. Nitrogen atom can attain an octet configuration by sharing three electrons with another nitrogen atom forming a triple bond three pairs of electrons shared Diagram of nitrogen bonding into octet configuration. Covalent bonds are of 3 types.
For ExFormation of chlorine HydrogenHydrogen chlorideMethane molecule etc. Methane which is a carbon atom with single covalent bonds to four hydrogen atoms is the simplest carbon compound. When two dissimilar nonmetals form bonds eg hydrogen and oxygen they will form a covalent bond but the electrons will spend more time closer to one type of atom than the other producing a polar covalent bond.
It shares its valence electrons with those of four H atoms. Double covalent bond. Carbon can form 4 single covalent bonds because it has 4 valence electrons and needs 4 more to fill its outer shell valence shell to form an octet.
One double and two single bonds. A carbon atom can form a single bond with 4 other carbons if those 4 carbons also form bonds with other atoms to fill their octets which can be seen in the structures of various organic compounds. Oxygen has 2 unshared pairs.
An atom that shares one or more of its electrons will. The electrons involved are in the outer shells of the atoms. When it bonds only with hydrogen it forms compounds called hydrocarbons.
The following diagram represents the structural formula of pentane C_3H_8. The structural formula of a water molecule is written. There are two pairs or 4 shared pairs of electrons between the atoms.
A single covalent bond is called a single bond or single covalent bond. The three major types of covalent bonds are single double and triple bonds. Other covalent bonds also have known bond lengths which are dependent on both the identities of the atoms in the bond and whether the bonds are single double or triple bonds.

2 7 Single Double And Triple Covalent Bonds Covalent Bonding Chemistry Education Chemistry Classroom
Single And Multiple Covalent Bonds Article Khan Academy
Covalent Bonding Ppt Video Online Download
Single And Multiple Covalent Bonds Article Khan Academy
Single And Multiple Covalent Bonds Article Khan Academy
Single Covalent Bond Definition And Examples
Single And Multiple Covalent Bonds Article Khan Academy
How To Distinguish Between Single Double And Triple Covalent Bonds Quora

Covalent Bond Chemical Bonding And Molecular Structure Chemistry Class 11

Single Covalent Bond Covalent Bonding Bond Chemistry
Single Covalent Bond Definition And Examples
Single Covalent Bond Definition And Examples





Posting Komentar untuk "4 Single Covalent Bonds"